For their studies on molecular dynamics, scientists in ICTP’s Condensed Matter and Statistical Physics (CMSP) section routinely use powerful computational techniques to simulate the structure and motion of atoms. These techniques can shed light on how fluids and solids interact at the atomic scale. Recently, CMSP scientists joined up with experimentalists at the University of Cambridge, UK, to use these techniques on a protein.
The Cambridge scientists, who study the physical origins of neurodegeneration (which can lead to diseases such as Alzheimer’s and Parkinson’s), wanted deeper insight into the fluorescence properties of a class of proteins known as amyloids that are associated with these diseases. Increased knowledge of these proteins’ glowing tendencies could have wide-ranging implications for everything from drug development to biosensing to biomolecular photonics devices. So they turned to ICTP’s molecular modellers for help.
Historically in biophysical chemistry, protein fluorescence has been associated with certain types of chemical groups called aromatic amino acids. But the amyloid proteins at the heart of the ICTP/Cambridge investigation can fluoresce with or without the aromatics. As ICTP biophysicist Ali Hassanali recalls, “The experimentalists came to us to ask, how do you rationalize this? This is what stimulated us to dig deeper into the problem.”
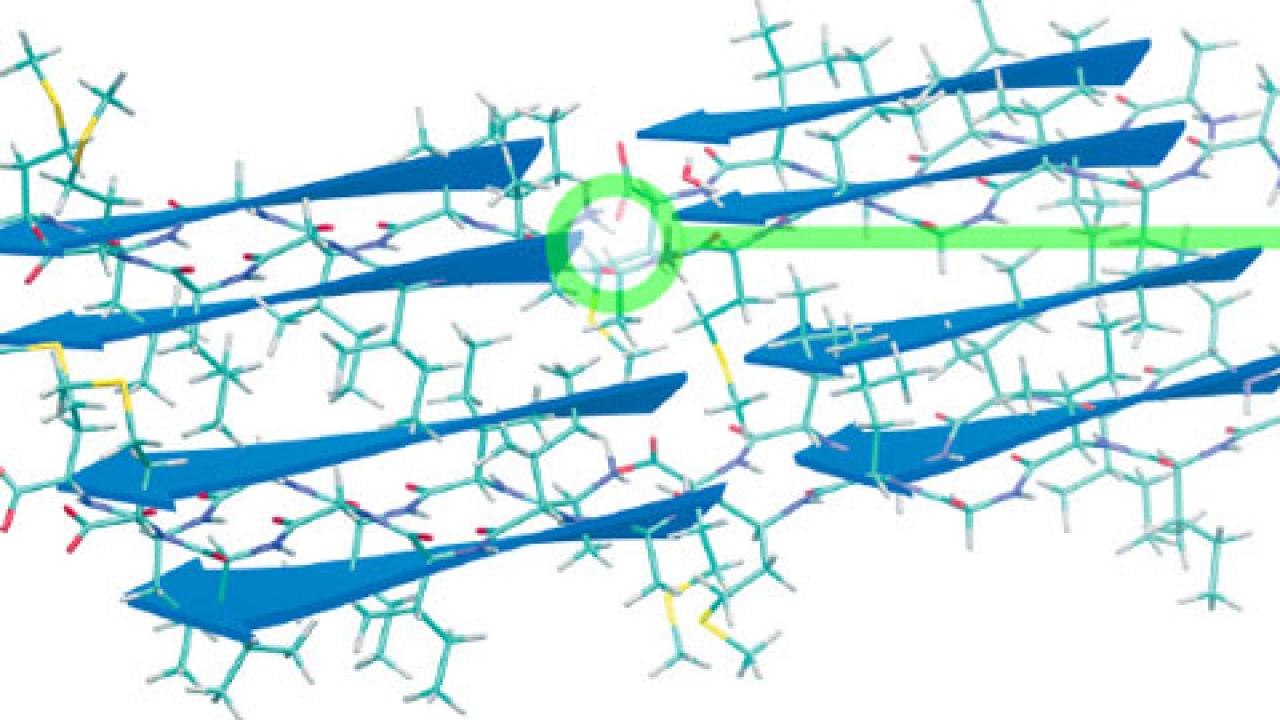
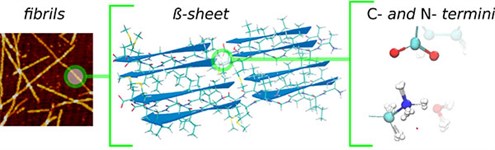 |
| Sketch of the hierarchical structure in protein fibrils. From left to right: fibrils to beta sheets and then finally to the strong hydrogen bonds between the N and C termini taken from a snapshot of a simulation where the protons are treated quantum mechanically, showing the delocalization of the proton. |
“Our goal was to do atomistic molecular modeling of the proteins and to study the optical properties,” explains Hassanali, who, together with ICTP physicist Ralph Gebauer and postdoctoral fellow Luca Grisanti spent months performing molecular dynamics simulations as well as determining optical spectra (work that required the use of high performance computers).
“The very peculiar thing that we found in these proteins is that protons can transfer from one tail of a protein to another, and this proton transfer appears to be strongly coupled to the interesting optical property. That is in short what we found,” says Hassanali.
The finding is significant, he says, and is sure to prompt a lot of interest in the community about trying to understand the physical chemistry of the problem in more detail.
The ICTP/Cambridge research was published in the 29 January 2016 online issue of the Journal of the American Chemical Society (DOI: 10.1021/jacs.5b11012).